Barbara Nsiah Anderson, PhD
Dermal matrices are commonly used to support tissue generation and vascularization in full-thickness skin defects. Numerous dermal matrices have been developed, ranging in composition from biological extracellular matrices to biosynthetic or synthetic polymers.
Additionally, several dermal matrix manufacturing processes have been employed to tune microarchitectural features such as porosity and pore size. The composition and microarchitectural features impact the body’s ability to interact with the dermal matrix and timeline needed to develop a vascularized wound bed capable of supporting a skin graft for definitive closure.
Dermal matrix microarchitecture and composition
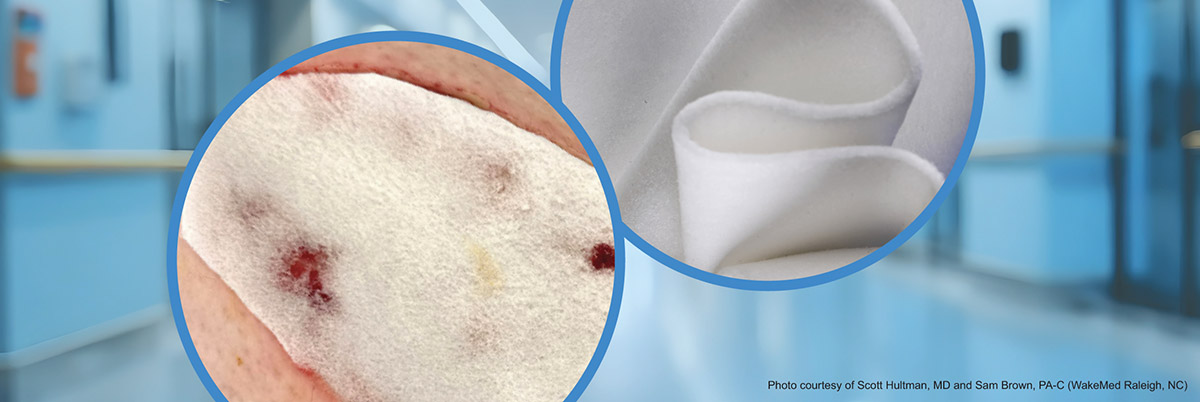
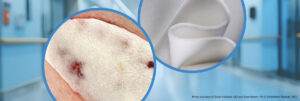
Each dermal matrix has a unique microarchitecture and composition. For example, Cohealyx® is a bioengineered, uniformly porous dermal matrix composed of bovine dermis, which contains collagen I and III (Figure A).1 Cohealyx allows for timely introduction of Type I and III collagen, which are natural biological cues that provide critical signaling during the wound healing process.2 Collagen is biocompatible, bioactive, and well conserved across species, making it an ideal material for a dermal matrix.3
Because collagen structure and sequence are well conserved amongst species, a dermal matrix such as Cohealyx is not expected to illicit an inflammatory response post implantation.1,3 In fact, Cohealyx-treated wounds demonstrated a lower immune cell filtration compared to other commercially available dermal matrices in a pre-clinical study.1
Scanning electron microscopy analysis reveals the range of pore sizes in a synthetic dermal matrix composed of biodegradable polyurethane foam, which has a porosity of 90% (Figure B).1 In contrast, the electrospun resorbable fiber matrix, depicted in Figure C, composed of polyglactin 910 and polydioxanone5 is less porous compared to Cohealyx (Figure A) and the biodegradable polyurethane foam dermal matrix (Figure B). A xenogeneic dermal matrix composed of fish skin contains smaller pores and has a lower porosity than Cohealyx (Figure D).6,7,8

A central role in wound bed readiness
Dermal matrices serve a central role in wound bed readiness. After implantation, dermal matrices fill the wound bed, provide structural support, scaffolding, and bioactive cues, including support of cellular infiltration and vascular tissue deposition important for the wound healing process. Immediately after implantation, host cells—including fibroblasts and endothelial cells—infiltrate the matrix, initiating collagen deposition and angiogenesis. Once the wound bed has developed sufficient vasculature, it is ready to graft.
The time required to develop a vascular bed capable of supporting a skin graft is highly dependent on the composition and microarchitecture characteristics of the dermal matrix. A comparison of several biological and biosynthetic dermal matrices with varying microarchitectures demonstrated differential in-vitro endothelial cell proliferation and angiogenic factor expression at 36 hours and in-vivo pre-clinical vascularization outcomes at 10 days.9
Early clinical data supports the pre-clinical finding that Cohealyx can support graft readiness in less than 14 days post implantation.10 Alternatively, other biological, biosynthetic, and synthetic dermal matrices have demonstrated wound readiness ranging from weeks to months.11-13
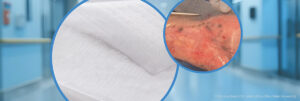
In pre-clinical work utilizing a full-thickness porcine model, wounds treated with Cohealyx demonstrated increased presence of endothelial cell infiltration compared to wounds treated with a fish skin graft.1 This data correlated with visual matrix observation at day seven: All Cohealyx treated wounds had >75% integration, whereas only 20% of fish skin graft treated wounds had this level of integration. When these wounds were grafted, the Cohealyx-treated wounds had 98% percent of graft take, whereas the fish skin graft wounds that were not fully ready for grafting only had 68% graft take.
Early clinical data supports the pre-clinical finding that Cohealyx can support graft readiness in less than 14 days post implantation.10 Alternatively, other biological, biosynthetic, and synthetic dermal matrices have demonstrated wound readiness ranging from weeks to months.11-13
As graft take and survival are driven by vascular support, the choice of dermal matrix plays a pivotal role in achieving good graft take outcomes and timely wound closure.
Dermal matrices: Key takeaways
Dermal matrix selection should consider microarchitecture, composition, integration time, and vascularization potential, as these directly affect time to graft. When early grafting is desired, matrices engineered for fast integration and rapid vascularization may offer a clinical advantage.
Looking for more articles about dermal matrices? Read Strategic Wound Closure in Resource-Limited Trauma: A Pragmatic Approach or Accelerating Healing for Complex Wounds: How Cohealyx™ Advances Readiness to Autografting.

About the author
Barbara Nsiah Anderson, PhD, Director of Scientific Affairs at AVITA Medical, leads pre-clinical study design and evaluation of new products for expansion of AVITA Medical’s acute wound care portfolio. She managed and designed the pre-clinical studies which supported the 2024 FDA clearance of Cohealyx®. She was instrumental in the successful publication of Cohealyx pre-clinical data in the Cureus journal.5 Dr. Anderson holds a PhD in bioengineering, is a fellow of the American Institute for Medical and Biological Engineering (AIMBE) and an inventor on several patents.
References
- Bush et al. Cureus Mar 31;17(3):e81517, 11
- Singh et al. Cariol Cardiovasc Med. 2023, 7:5-16
- Gallo et al. J Funct. Biomater. 2020, 11,79.
- Wagstaff et al. Eplasty 2015, 24:15:e13
- Lo et al. Burns 2022, 48(3):529-538
- MacEwan et al. Cureus, 2017, 9(8):e1614
- Magnusson et al. Mil Med. 2017;182(S1):383-388
- Data on file: publication in progress
- Agostinis et al. Biomedicines 2021, 9:1458
- Akpunonu et al J. Surg 2025, 10:11337
- Chowdry et al. Eplasy 2024 Jan 4:24:e2.
- Mittal et al. Burns Open 2024, 8(3):220-227
- Fruergaard et al. Burns Open 2025, 9 100378